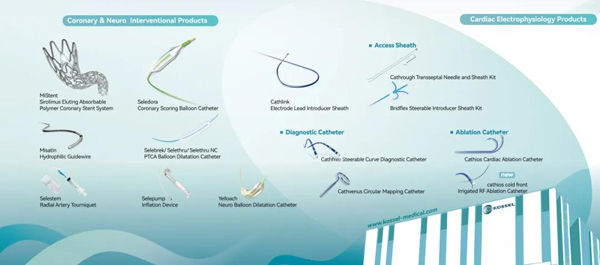
January 20, 2026 – The strategic cooperation signing ceremony between Kossel Medtech (Suzhou) Co., Ltd (“Kossel”) and Medtronic (Shanghai) Management Co., Ltd. (“Medtronic”) was successfully held. This collaboration marks the establishment of a deep strategic partnership. Centered on Kossel’s core product, the Seledora® Coronary Scoring Balloon Catheter, the two sides will work together to promote its clinical application and benefit more patients with coronary artery disease.

Senior leaders from both companies attended the ceremony, including Mr. Gu Yushao, Sr. Vice President, President of Greater China; Mr. Hu Qing, Founder and CEO of Kossel; Mr. Tao Jia, VP China CRDN; Ms. Pan Xingzhen, Executive President of Kossel; and Mr. Zhou Quan, VP of Strategy & BI and Partnerships & Digital Innovation, etc, and witness this grand event of industry together.
Strategic Synergy: Connecting Local Innovation power with Global Resources
This partnership represents a deep integration of local innovative strength and global medical technology leadership, bringing new momentum to coronary intervention.
Kossel, as a local innovator in cardiovascular intervention, focuses on developing high-end medical devices based on clinical needs. Its self-developed Seledora® Coronary Scoring Balloon offers a precise solution for complex lesions like moderate-to-severe calcification. Medtronic, a global leader, brings its extensive nationwide network, professional clinical promotion system, and deep industry resources in China to support the widespread adoption of this innovative product.

Both parties believe this collaboration is a key step for the commercialization of Kossel’s product and an important move for Medtronic’s localization strategy. By combining strengths, we aim to accelerate the introduction of valuable clinical solutions and help raise treatment standards.
Co-creating the Future: Building an Innovation Ecosystem for Healthy China
This collaboration is strategically significant for both. Kossel will leverage Medtronic’s comprehensive system to rapidly increase the accessibility of Seledora® scoring balloons. Medtronic, by partnering with the locally-engaged innovator Kossel, aims to better integrate global experience with Chinese practice, respond faster to clinical needs, improve solutions for complex patients, and promote standardized, sustainable development in coronary therapy.

Leadership Perspectives:

Mr. Gu Yushao: “Chinese healthcare is evolving towards quality, stability, and sustainability. Medtronic is committed to fostering local innovation. Partnering with Kossel is a win-win to create a pathway tailored to China’s needs and deliver long-term clinical value.”

Mr. Hu Qing: “Addressing complex coronary cases drives our innovation. True innovation requires not just technology, but also standardized application and promotion, which needs a mature system’s support. Partnering with Medtronic, who shares our patient-centric and long-term vision, is a natural step to advance local innovation and release greater clinical value.”

Mr. Tao Jia: “The growing scale and complexity of cases require a new stage of precise, multi-device therapy. No single technology suffices. Partnering with Kossel allows us to combine global experience with local practice, improving solutions for complex patients and promoting standardized, sustainable development.”

Ms. Pan Xingzhen: “The Seledora® balloon catheter is built on a solid foundation of R&D, quality, and clinical support. This collaboration focuses not only on the product but also on aligning our working mechanisms. Kossel will continue to develop mature products, complementing Medtronic’s strengths to offer richer, more reliable solutions.”
Both parties will establish comprehensive collaboration mechanisms across product promotion, academic exchange, and clinical research. By integrating R&D innovation with commercial expertise, we aim to build a better coronary intervention ecosystem, advance cardiovascular disease treatment, and contribute to the “Healthy China” initiative.